CDMO Services
Vertically integrated CDMO services for oligonucleotide and mRNA therapeutics
From raw materials to GMP drug product: Fewer handoffs, faster timelines, greater confidence
Hongene simplifies complex supply chains
By combining raw material production with advanced oligonucleotide and mRNA CDMO services. This vertically integrated model reduces handoffs, accelerates timelines and ensures regulatory alignment at every stage of development.

Our Services
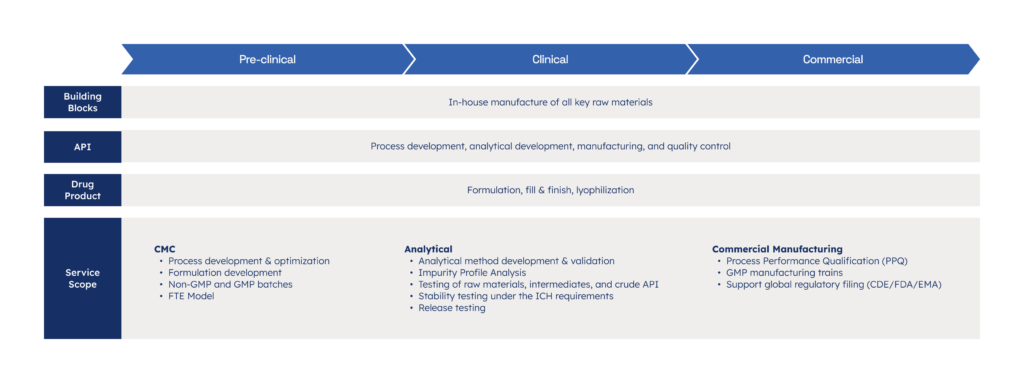
Oligonucleotide CDMO
From early development to tonne-scale GMP production, we support complex oligonucleotide programs with speed and precision.
- Process and analytical development
- Chemoenzymatic ligation for scalable, high-purity siRNA, sgRNA and other oligonucleotides
- GMP production with up to tonne scale
- QC, regulatory support, and release testing
mRNA CDMO
Complete in-house control of mRNA raw materials — from enzymes to lipids — through to GMP manufacture and fill–finish.
- Plasmid DNA template generation and linearization
- One-pot in vitro transcription (IVT) (integrated plasmid linearization and transcription)
- Capping with HiXCap™ for improved translation efficiency
- Purification and QC (≥98% full-length, dsRNA <0.01%)
- LNP formulation, lyophilization, aseptic fill–finish
Custom synthesis
- Custom-built raw materials for RNA innovation
- Core building blocks (standard and custom phosphoramidites)
- Modified chemistries (ribose, backbone, nucleobase, conjugates)
- Specialty reagents (HiXCap™, linkers, novel monomers)
- Rapid turnaround for bespoke requests